SHOP BY NEED
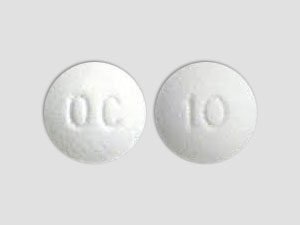
Product Description: OxyContin OC 20mg
Overview:
OxyContin OC 20mg is a prescription extended-release opioid medication used for the management of severe, chronic pain that requires continuous, long-term treatment. Each tablet contains 20mg of oxycodone hydrochloride, designed to provide consistent pain relief over a 12-hour period. The OC (OxyContin Controlled) formulation includes abuse-deterrent properties that make the tablet difficult to crush, break, or dissolve, helping to prevent misuse and abuse.
Key Features:
-
Active Ingredient: Oxycodone Hydrochloride 20mg
-
Form: Extended-release oral tablet
-
Use: For opioid-tolerant patients with severe chronic pain
-
Dosing: Taken twice daily, every 12 hours
-
Abuse-Deterrent: Designed to deter tampering and misuse
How It Works:
Oxycodone binds to opioid receptors in the brain and spinal cord, altering the perception and response to pain. The extended-release formulation allows for a steady, controlled release over 12 hours, maintaining effective pain control.
Dosage & Administration:
-
Intended only for opioid-tolerant individuals
-
Typical dosage: 20mg every 12 hours, as prescribed by a healthcare provider
-
Tablets must be swallowed whole; do not crush, chew, or break
-
May be taken with or without food
-
Follow all medical guidance strictly to avoid overdose
Warnings & Precautions:
-
High risk of addiction, abuse, and misuse
-
Risk of life-threatening respiratory depression
-
Accidental ingestion, especially by children, can be fatal
-
Use with caution if you have respiratory, liver, or kidney problems
-
May cause neonatal opioid withdrawal syndrome if used during pregnancy
-
Avoid alcohol and CNS depressants during use
Common Side Effects:
-
Constipation
-
Nausea
-
Drowsiness
-
Dizziness
-
Dry mouth
Serious Side Effects:
-
Respiratory depression
-
Severe sedation
-
Allergic reactions
-
Signs of overdose (slow breathing, loss of consciousness)
Storage:
-
Store at room temperature in a secure location
-
Keep out of reach of children and unauthorized persons
-
Dispose of unused medication properly via drug take-back programs
Legal Classification:
-
Schedule II controlled substance due to high abuse potential